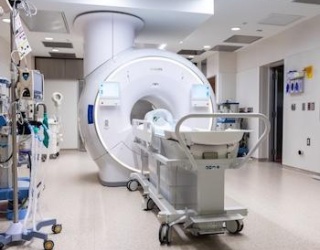
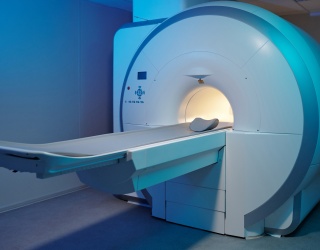
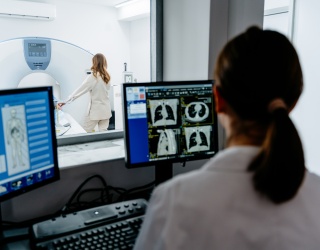
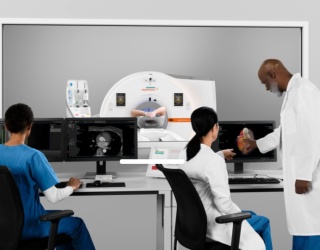
The update was shared in connection with the AANS Annual Meeting in San Antonio (May 1–4, 2026). Esaote positions I-Genius as its first dedicated intraoperative MRI system for neurosurgical brain tumor procedures, designed to bring MRI verification steps into the surgical suite instead of treating imaging as an external detour.
In glioma surgery, the margin between tumor and functional tissue can be difficult to define, and the goal of maximal safe resection often has to be balanced against preserving neurological function. Teams routinely use tools such as neuronavigation, mapping techniques, and—in selected cases—awake procedures to reduce risk. Even so, a core limitation remains: once the operation is underway, anatomy can shift and visual cues are indirect, making it hard to know in real time whether residual tumor remains. I-Genius is presented as a practical way to re-check the situation repeatedly—supporting decisions about whether to continue resection, adjust the approach, or stop—based on new intraoperative MRI acquisitions.
Why low-field MRI can be advantageous in the surgical suite
In diagnostic neuroradiology, most routine brain MRI is performed at 1.5T or 3T, where image quality and protocol breadth are well established. Inside the operating room, however, the primary constraints are different: physical access to the patient, sterility, anesthesia and monitoring logistics, and the need to keep the case moving. In that environment, low-field MRI can be advantageous not because it competes directly with high-field image quality, but because it can support an “OR-first” design that fits the realities of surgery.
Open, low-field architectures can make it easier to maintain surgical access around the head and to integrate imaging into the room without forcing a major redesign of the procedure. For glioma cases in particular, the clinical value is tightly tied to workflow: if imaging requires moving the patient out of the suite, breaking the sterile field, or introducing complex handoffs, teams may lose time—and the usefulness of “check-and-adapt” imaging diminishes. A low-field system designed for intraoperative use aims to reduce these disruptions, making multiple re-checks more feasible over the course of a single case.
That said, low-field remains a trade-off. Lower signal-to-noise ratio generally means teams must balance resolution, scan time, and protocol scope. The operational question is whether the imaging is sufficiently informative to change intraoperative decisions—confirming suspected residual tumor, differentiating tumor from expected postoperative change, or clarifying anatomy after brain shift—without slowing the procedure or adding fragile dependencies on scarce staff and specialized equipment.
What Esaote emphasizes with I-Genius
Esaote’s core message with I-Genius is repeatable intraoperative verification with minimal disruption. The company describes a workflow in which the patient remains on the same table throughout the procedure, allowing teams to perform multiple MRI checks without repositioning. In practical terms, the concept targets two recurring pain points in intraoperative imaging: reducing the complexity of transfers and preserving momentum in the surgical suite while maintaining a safety-focused environment.
The accompanying materials also underscore surgical integration: imaging as an embedded step in the procedure, rather than a separate event that forces a pause and reset. For teams, the day-to-day implications are less about a single scan and more about how reliably the system can support repeated cycles of resection, imaging, interpretation, and next-step decisions.
What to watch next in European practice
For European centers considering intraoperative MRI—especially low-field concepts—the deciding factors are usually practical and multidisciplinary. Sites will look closely at how the system fits into existing surgical suites, what MR safety zoning and training look like in daily practice, and how anesthesia, monitoring, and MR-compatible equipment will be handled without bottlenecks. Radiology and OR leaders will also focus on how images move through the hospital ecosystem: documentation, storage, reporting, and alignment with PACS and the medical record.
Clinically, adoption will hinge on how teams define success in glioma surgery and whether intraoperative MRI can deliver it consistently: improved confidence about extent of resection, fewer early re-operations, preservation of function, and overall efficiency in the surgical suite. The key test will be whether repeat imaging becomes a routine, dependable part of glioma workflows—not an occasional capability that is too disruptive to use when it matters most.